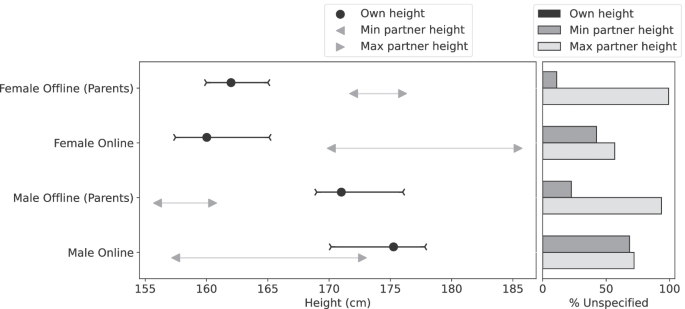 
Secondary outcomes included age at diabetes diagnosis and safety (adverse events).Īmong 2159 newborn infants (1021 female ) who were randomized, 1744 (80.8%) completed the trial. Primary outcome was type 1 diabetes diagnosed according to World Health Organization criteria. The minimum duration of study formula exposure was 60 days by 6 to 8 months of age.  
The participants received either a casein hydrolysate or a conventional adapted cow's milk formula supplemented with 20% of the casein hydrolysate. The follow-up of the participants ended on February 28, 2017. To test the hypothesis that weaning to an extensively hydrolyzed formula decreases the cumulative incidence of type 1 diabetes in young children.Īn international double-blind randomized clinical trial of 2159 infants with human leukocyte antigen-conferred disease susceptibility and a first-degree relative with type 1 diabetes recruited from May 2002 to January 2007 in 78 study centers in 15 countries 1081 were randomized to be weaned to the extensively hydrolyzed casein formula and 1078 to a conventional formula. There are no intact proteins in extensively hydrolyzed formulas. Early exposure to complex dietary proteins may increase the risk of type 1 diabetes in children with genetic disease susceptibility.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
